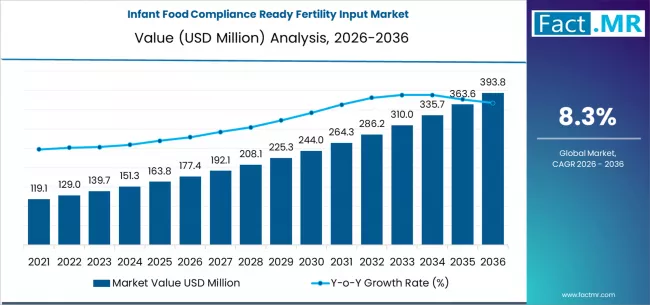
According to Fact MR’s latest analysis, the France Infant Food Compliance-Ready Fertility Input Market is valued at USD 420 Million in 2026, projected to reach USD 455 Million in 2027, and expected to grow to USD 890 Million by 2036, registering a CAGR of 6.9%. The market is set to create an incremental opportunity of USD 470 Million over the forecast period.The market is undergoing transformation driven by stringent EU food safety regulations, rising fertility awareness, and increasing demand for scientifically formulated infant nutrition products. France’s strong regulatory ecosystem and premium dairy infrastructure position it as a leader in compliance-ready infant nutrition inputs.
Quick Stats
- Market Size (2026): USD 420 Million
- Market Size (2027): USD 455 Million
- Forecast Value (2036): USD 890 Million
- CAGR (2026–2036): 6.9%
- Incremental Opportunity: USD 470 Million
- Leading Segment: Nutritional Ingredients for Infant Formula (~52% share)
- Leading Country: France
- Key Players:Danone S.A., Lactalis Group, Nestlé S.A.
Executive Insight for Decision Makers
France’s market is shifting toward compliance-first, science-backed infant nutrition ecosystems.
- Manufacturers must invest in regulatory-compliant, traceable ingredients
- Investors should focus on premium and specialized nutrition segments
- OEMs need to align with EU food safety and fertility health standards
Risk: Non-compliance or lack of innovation may lead to market exclusion due to strict regulatory frameworks.Market Dynamics
Key Growth Drivers
- Increasing fertility awareness and maternal health focus
- Strong demand for premium infant nutrition products
- Strict EU and French food safety regulations
- Growth in functional and fortified ingredients
Key Restraints
- High regulatory compliance costs
- Complex approval processes for new ingredients
- Price sensitivity in mass-market segments
Emerging Trends
- Rise in organic and clean-label infant food inputs
- Increasing demand for plant-based and alternative proteins
- Growth of precision nutrition and functional ingredients
- Expansion of traceability and transparency systems
Segment Analysis
- Leading Segment: Infant formula nutritional inputs (~52% share)
- Fastest-Growing Segment: Functional fertility-support ingredients
Breakdown
- Product: Vitamins, minerals, proteins, probiotics
- Application: Infant formula, baby food, supplements
- End-use: Infant nutrition manufacturers
Strategic Importance: High-value ingredients drive premiumization and compliance differentiation in France.
Supply Chain Analysis (Critical Insight)
- Raw Material Suppliers: Dairy producers, plant-based ingredient suppliers
- Ingredient Manufacturers: Specialized nutrition companies (e.g., Danone S.A.)
- Processors: Infant food manufacturers
- Distributors: Retail chains, pharmacies, online platforms
- End-Users: Parents, healthcare providers
Flow Explanation:
Dairy/plant suppliers → Ingredient processors → Infant food manufacturers → Retail/pharma → ConsumersFrance’s supply chain emphasizes traceability, quality assurance, and regulatory compliance.Pricing Trends
- Premium pricing dominates due to high compliance and quality standards
- Influencing factors:
- Regulatory certifications
- Ingredient sourcing (organic, specialty)
- R&D and formulation complexity
Margin Insight: Premium and functional ingredients command higher margins due to health positioning.
Regional Analysis
Top Countries (Europe Context)
- France – 6.9% CAGR (regulatory leadership)
- Germany – ~6.5% CAGR (strong demand)
- UK – ~6.3% CAGR (premium products)
- Italy – ~6.1% CAGR (nutrition demand)
- Spain – ~6.0% CAGR (emerging growth)
Insights
- France leads in compliance-driven innovation
- Western Europe focuses on premiumization
- Emerging regions emphasize affordability and access
Competitive Landscape
The market is moderately consolidated, dominated by global nutrition and dairy companies.Key Players
- Danone S.A.
- Lactalis Group
- Nestlé S.A.
- Arla Foods
- FrieslandCampina
Strategies
- Investment in R&D and clinical nutrition
- Expansion of organic and clean-label portfolios
- Focus on regulatory compliance and certifications
- Strategic partnerships with healthcare providers
Strategic Takeaways
For Manufacturers
- Focus on compliance-ready and premium ingredients
- Invest in traceability and quality systems
For Investors
- Target high-margin specialized nutrition segments
- Focus on innovation-driven companies
For Distributors
- Emphasize premium branding and trust factors
- Expand presence in pharmacy and online channels
Future Outlook
France’s market is expected to evolve into a highly regulated, innovation-driven nutrition ecosystem.
- Growth in functional and fertility-support ingredients
- Increasing adoption of precision nutrition technologies
- Strong push toward sustainability and clean-label products
Long-term opportunity lies in science-backed, personalized infant nutrition solutions.
Conclusion
France stands at the forefront of compliance-ready infant nutrition innovation, driven by its regulatory strength and premium product ecosystem.Companies that align with quality, transparency, and scientific advancement will unlock significant growth opportunities in this evolving market.
Why This Market Matters
This market plays a critical role in early-life nutrition and maternal health, making it a cornerstone of future healthcare and food innovation.