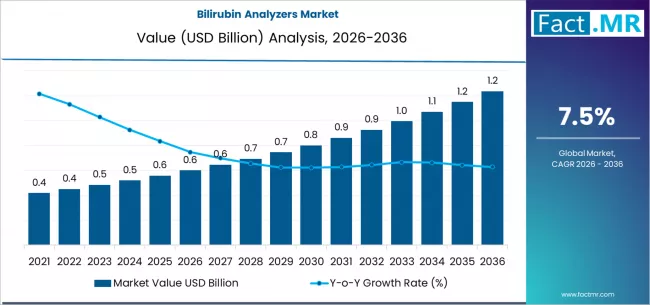
The global landscape for neonatal diagnostics is undergoing a significant shift toward non-invasive, high-precision monitoring. According to a new comprehensive industry analysis, the Bilirubin Analyzers Market is set to double in value, growing from USD 0.6 billion in 2026 to USD 1.2 billion by 2036.
This steady growth, moving at a CAGR of 7.5%, reflects a worldwide prioritization of infant health and the integration of automated diagnostic protocols within clinical environments. As healthcare providers seek to balance diagnostic accuracy with patient comfort, the market is seeing a definitive move toward technologies that offer rapid results without the need for frequent blood sampling.
Transcutaneous Meters: Setting a New Standard in Patient Comfort
The traditional method of serum analysis is increasingly being supplemented—and in many cases, replaced—by advanced bedside technology. The Transcutaneous Bilirubin Meters segment is projected to hold a dominant 63.8% share of the market by 2026.
These devices allow for instantaneous, non-invasive screening, which is critical in neonatal care. By eliminating the stress of needle sticks for newborns, healthcare facilities are achieving higher operational efficiency while meeting stringent regulatory adherence standards for patient monitoring.
At a Glance: Key Market Statistics
-
2026 Estimated Market Value: USD 0.6 Billion
-
2036 Projected Market Value: USD 1.2 Billion
-
Forecast CAGR (2026-2036): 7.5%
-
Dominant Care Setting: Hospitals and NICUs (68.2% Share)
-
Leading Technology: Transcutaneous Bilirubin Meters (63.8% Share)
Hospitals and NICUs: The Frontline of Diagnostic Innovation
Hospitals and Neonatal Intensive Care Units (NICUs) remain the primary engine for market growth, accounting for 68.2% of the application share. Facility operators are currently optimizing equipment selections to support:
-
Advanced Diagnostic Protocols: Standardizing screening to ensure early detection of jaundice.
-
Compliance & Cost Reduction: Utilizing multi-parameter compatibility systems to lower the total cost of care.
-
Operational Control: Investing in automated configurations that minimize manual data entry and human error.
Regional Growth Engines: India and China Lead the Pace
While North America and Europe remain high-value markets, the fastest expansion is occurring across the Asia Pacific region, where healthcare infrastructure is scaling rapidly.
-
India: Leading global growth with a projected 9.2% CAGR, fueled by massive investments in maternal and child health infrastructure.
-
China: Following closely at an 8.6% CAGR, driven by an emphasis on healthcare excellence and the integration of high-precision diagnostic tools.
-
Indonesia & Brazil: Both regions show robust potential with CAGRs of 7.8% and 7.6% respectively, as modern healthcare applications become more accessible to broader populations.
Competitive Dynamics and Key Players
The competitive landscape is defined by a blend of medical technology giants and specialized diagnostic innovators. These companies are currently prioritizing “diagnostic heritage” and innovation in microfluidic assays and optical spectroscopy to maintain market leadership.
Key industry participants include:
-
Dräger (Leader in integrated diagnostic automation)
-
Konica Minolta
-
Philips
-
Natus Medical
-
GE HealthCare
-
Siemens Healthineers
-
Roche
-
Abbott
-
Mindray
-
Radiometer
Strategic Insights for Healthcare Decision Makers
For clinical directors and healthcare administrators, the shift toward transcutaneous technology represents a strategic opportunity to improve throughput in NICUs while enhancing the quality of care. Understanding the transition from bench-top serum analyzers to handheld, point-of-care solutions is essential for future-proofing diagnostic departments.