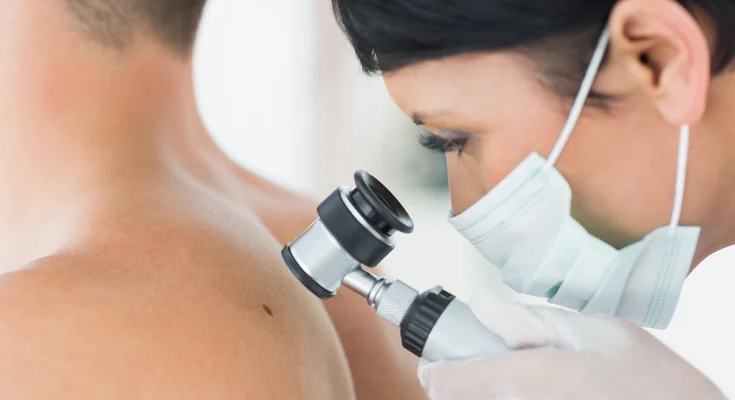
The study on the Global Dermatology CRO Market Insights published by Fact.MR is a comprehensive analysis of the key factors that are likely to determine the growth of Dermatology CRO Market Insights in the upcoming years. Further, the study dives deep to investigate the micro and macro-economic parameters that are expected to influence the global scenario of the Dermatology CRO Market Insights during the forecast period (2021-2031).
The report examines the current trends, growth opportunities, restraints, and market drivers that are projected to influence the overall dynamics of the Dermatology CRO Market Insights in the assessment period.
Request Sample https://www.factmr.com/connectus/sample?flag=S&rep_id=7178
Dermatology CRO Market Insights Segmentation
-
- By Type :
- Drug Discovery
- Target Validation
- Lead Identification
- Lead Optimization
- Clinical
- Phase I
- Phase II
- Phase III
- Phase IV
- Preclinical
- Drug Discovery
- By Service :
- Project Management/Clinical Supply Management
- Data Management
- Regulatory/Medical Affairs
- Medical Writing
- Clinical Monitoring
- Quality Management/Assurance
- Bio-statistics
- Investigator Payments
- Laboratory
- Patient and Site Recruitment
- Technology
- Other Services
- By Region :
- North America
- Latin America
- Europe
- APAC
- MEA
- Drug Discovery
- Target Validation
- Lead Identification
- Lead Optimization
- Clinical
- Phase I
- Phase II
- Phase III
- Phase IV
- Preclinical
- By Type :
Competitive Analysis
According to the market research analysis, partnerships and expansion have been the prominent development in Dermatology CRO market in recent years. IQVIA HOLDINGS INC., Covance Inc., Pharmaceutical Product Development, LLC (PPD), Parexel International Corporation, Charles River Laboratories International, Inc., Icon, Plc, Medidata Solutions, Inc., Syneos Health, Pharmaron, GVK Biosciences Private Limited, Wuxi AppTec, MEDPACE HOLDINGS, INC., PRA Health Sciences, CTI Clinical Trial & Consulting, Bioskin, Proinnovera, Biorasi, Javara, TFS are the prominent providers of Dermatology CRO services.
Some of the recent developments of key Dermatology CRO providers are as follows:
- In 2021 Thermo Fisher signed a binding deal to buy PPD for US$ 17.4 Bn. This purchase will greatly strengthen Thermo Fisher’s position in the clinical research services market, particularly in the early phase clinical trials business. In addition to strengthening their services, healthcare contract research businesses are aiming to provide their services on a worldwide basis.
- In February 2021, Icon plc signed a binding deal to buy PRA Health Sciences for a valuation of around US$ 12 Bn. Customers will benefit from the merged firm because of the enhanced geographic, functional, and therapeutic scale.
- In October 2019, Parexel strengthened its ties with the Society for Clinical Research Sites (SCRS), to safeguard the interests of clinical research sites. The company implemented many significant initiatives as a Site Engagement Partner targeted at enhancing the patient experience in clinical trials.